The researchers retrospectively reviewed data from a large national managed-care network, specifically 340,372 enrollees who received an initial diagnosis of acute conjunctivitis from 2001 through 2014, seeking those who had filled one or more antibiotic prescriptions within 14 days of diagnosis. They also analyzed those patients’ demographic characteristics and medical information.
Nakul Shekhawat, MD, MPH, of the University of Michigan’s Kellogg Eye Center, and colleagues found that of those 340,372 patients newly diagnosed with pinkeye during the period studied, 198,462 (58 percent) filled at least one topical antibiotic prescription within two weeks of their diagnosis. Twenty percent of that cohort (38,774) filled scripts for combined antibiotic-corticosteroid drops, which may worsen and prolong the course of viral conjunctivitis. Further analysis revealed that the more affluent and educated the conjunctivitis patient was, the likelier it was that they’d fill a prescription. There was no relationship between filling antibiotic scripts and medical risk factors such as contact lens wear, HIV infection or AIDS.
Compared with patients first diagnosed by ophthalmologists, patients initially diagnosed with pinkeye by the following types of providers displayed higher odds ratios with 95-percent confidence intervals of filling antibiotic scripts: optometrist (OR, 1.26; 95% CI, 1.21–1.31); urgent-care physician (OR, 3.29; 95% CI, 3.17–3.41); internist (OR, 2.79; 95% CI, 2.69–2.90); pediatrician (OR, 2.27; 95% CI, 2.13–2.43); or family practitioner (OR, 2.46; 95% CI, 2.37–2.55).
Inability to quickly determine the etiology of a patient’s conjunctivitis
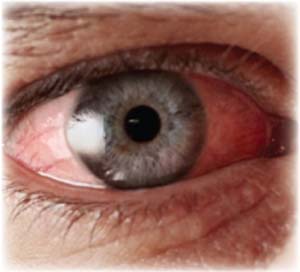 |
A lack of patient education may lead patients to believe that they need antibiotics to treat every case of conjunctivitis. Another possible culprit in pediatric patients is school and daycare policies excluding children with untreated acute conjunctivitis.
Dr. Shekhawat notes that spreading the word to providers outside the eye care community is complicated by the fact that other providers see the majority of acute conjunctivitis cases (83 percent in the study). “Getting the message out is difficult because non-eye care providers are unlikely to be exposed to communications from the American Academy of Ophthalmology or similar organizations, and are far more likely to get the message if it comes from their own medical organizations,” he says. “The AAO has taken part in the national Choosing Wisely campaign and discouraged indiscriminate use of antibiotics for acute conjunctivitis, but I doubt that message has permeated beyond the eye-care community. General medical societies such as the American Academy of Pediatrics should communicate with ophthalmologists to produce and disseminate consensus statements regarding the management of conjunctivitis.”
Regarding the role of school and daycare policies in the overprescribing, Dr. Shekhawat says that state health departments vary in their guidance on kids with acute conjunctivitis. “Some states’ policies are far more nuanced and evidence-based than others. States with overly restrictive policies should formulate clear, evidence-based guidelines that discourage indiscriminate antibiotic use,” he says.
“There are no quick solutions to the antibiotic overuse identified in our study,” Dr. Shekhawat adds. “However, the policy approaches discussed above are likely to have the greatest impact, since they address some of the root causes of the problem.”
1. Shekhawat NS, Shtein RM, Blachley TS, Stein JD. Antibiotic prescription fills for acute conjunctivitis among enrollees in a large United States managed care network. Ophthalmology. 2017; doi: 10.1016/j.ophtha.2017.04.034. [Epub ahead of print]
2. Udeh B, Schneider J, Ohsfeldt R. Cost effectiveness of a point-of-care test for adenoviral conjunctivitis. Am J Med Sci. 2008;336:3:254-264.
Dextenza FDA Update
In a Complete Response Letter to Ocular Therapeutix regarding its resubmission of a New Drug Application for Dextenza (dexamethasone insert) 0.4 mg for the treatment of ocular pain following ophthalmic surgery, the U.S. Food and Drug Administration stated it could not approve the NDA in its present form. The CRL referred to deficiencies in manufacturing processes and analytical testing related to the manufacture of drug products identified during a pre-NDA approval inspection of the Ocular Therapeutix manufacturing facility in May 2017. FDA raised no safety or efficacy issues with the drug. REVIEW



