Lead author Rebekah A. Braslow, MD, of the Pritzker School of Medicine, University of Chicago and Division of Ophthalmology, NorthShore University Health System in Illinois, and colleagues undertook the study to find out what impact the American Academy of Ophthalmology’s 2011 HCQ dosing guidance2 has had on prescribing rheumatologists, and to help predict the long-term impact of the 2016 revision3 of these guidelines. In 2011, the AAO recommended that patients receive no more than 6.5 mg of HCQ per kilogram of ideal body weight daily; in 2016, that recommendation was revised to 5 mg/kg/day based on actual measured body weight. Dr. Braslow and colleagues found, however, that many patients in the health-care system they studied continued to be prescribed HCQ at levels that may pose an undue risk to their retinal
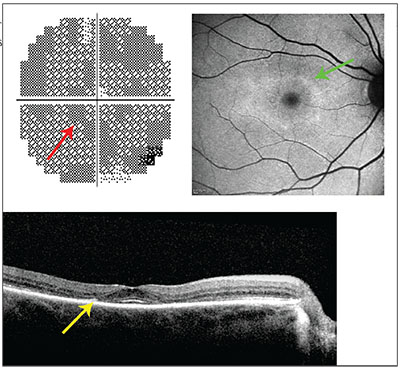 |
| Paracentral scotomata (top left, red arrow) in a 10-2 visual field test is a sign of toxic hydroxychloroquine dosing, as is an annulus of hyperautofluorescence (top right, green arrow) on blue fundus autofluorescence imaging. Other signs to look for are paracentral loss of the ellipsoid line and thinning of the outer nuclear layer (bottom, yellow arrow). |
The researchers performed a retrospective review of the medical records of the NorthShore University Health System. They identified 554 patients who’d been prescribed HCQ at least once between 2009 and 2016, and who’d also been seen by a staff ophthalmologist.
Ninety-two patients had been started on HCQ before the 2011 dosing guidelines; 462 started taking it after them. The researchers found that 54.3 percent of patients who started HCQ before the 2011 recommendations were exceeding the 2011 dosing threshold of 6.5 mg/kg/day per ideal body weight, compared with 49.4 percent who started HCQ therapy after the 2011 guidance. When the researchers applied the 2016 dosing guidelines of up to 5 mg/kg/day per actual weight using patient weights obtained from patient records, they found that 56 percent of the 527 patients still on HCQ therapy were getting excess doses. Many of the excess doses were within 50 mg of the lower dosing threshold established in 2016; but 43 percent of the current HCQ patients (224 of 527) were getting doses more than 50 mg/day above the recommended dose—some up to 450 mg/day more than the threshold amount.
The prevalence of retinal toxicity arising from HCQ is not insubstantial, with a 2014 study pegging it at 7.5 percent overall in patients on HCQ for a minimum of five years, spiking to nearly 20 percent after 20 years of daily therapy.4 The retinal damage and vision loss that accompanies HCQ toxicity is irreversible and progresses for a period of time after discontinuing the drug. This complication leaves doctors and their patients with no recourse other than ophthalmologic monitoring of retinal toxicity once it develops, and the dilemma of whether to discontinue HCQ to slow and eventually arrest retinopathy and vision loss. Prior to modern screening techniques, a characteristic bull’s-eye pattern of maculopathy was the first definitive sign of retinal toxicity, but it signifies later-stage damage to the RPE and photoreceptors. With spectral-domain optical coherence tomography, multifocal electroretinography and fundus autofluorescence imaging, more subtle early abnormalities are detectable.
Because the 2016 dosing threshold is based solely on a patient’s weight instead of an ideal weight calculated using height, the authors note that the likelihood of a “safe” dose of HCQ increases correspondingly with weight, and that weight-based dosing reduces the disproportionate risk of excess dosing to thin patients. The authors suggest that a “screening referral” to an ophthalmologist isn’t necessary when initiating therapy in patients, and that “the creative use of EMR to guide proper dosing” would be more helpful to non-ophthalmologists in determining a safe starting dose.
The American College of Rheumatologists released a position paper5 in 2016 subsuming much of the AAO’s latest guidance. According to Michael F. Marmor, MD, professor of ophthalmology, Byers Eye Institute, Stanford University, “The biggest problem is educating rheumatology. I have published a few alerts in rheumatology publications, but even their subspecialty journals have not embraced major editorial notice to the discipline.” Dr. Marmor, who was not involved in the current study on HCQ dosing patterns, is the lead author of the AAO’s 2011 HCQ dosing guidelines and the 2016 revision, and lectures widely on the topic of retinal toxicity.
He notes that rheumatology “is gradually coming around, teaching these new recommendations to trainees and educating the field.” Dr. Marmor also thinks there is room for improvement on this topic in ophthalmology, however, adding, “Ophthalmologists often fail to follow these guidelines, so our field bears some responsibility for educating itself.”
To step up HCQ dosing surveillance among all health-care providers, the authors recommend system-wide education and EMR-generated prompts and alerts to highlight potentially risky dosing. Dr. Marmor thinks that the idea of working warnings and dosing templates into EMR has merit, but it’s not always feasible. “Alerts only work in systems that accept them. Some EMR is too complex, and notices are hard to get accepted in large academic systems. Also remember that every drug has problems, and if 45 drug alerts pop up every time we see a patient, the effect will be to ignore them,” he warns.
The study authors identify the commercially available form of HCQ (Plaquenil) as another contributor to excess dosing, since it only comes in 200-mg pills. They propose alternate-day regimens and the use of compounders as ways to fine-tune dosing. “A 50-mg tablet would be great,” says Dr. Marmor, “except that there is no incentive for the drug industry to change an old drug.” But he agrees that alternate-day doses are a simple solution. “That is a mild annoyance, but not an excuse to give an incorrect dosage,” he says.
 |
“We are trying to expand awareness, and in our field of ophthalmology, we all have an individual responsibility to inform our colleagues in other fields about HCQ use, as well as about many other diseases that impact the eye,” he says. REVIEW
1. Braslow RA, Shiloach M, Macsai MS. Adherence to hydroxychloroquine dosing guidelines by rheumatologists. Ophthalmology. 2017 Jan 30. pii: S0161-6420(16)31143-5. doi: 10.1016/j.ophtha.2016.12.021. [Epub ahead of print] PMID: 28153440
2. Marmor MF, Kellner U, Lai TY, et al, American Academy of Ophthalmology. Revised recommendations on screening for chloroquine and hydroxychloroquine retinopathy. Ophthalmology 2011;118:415–22.
3. Marmor MF, Kellner U, Lau TY, et al, American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology 2016;123:1386-94.
4. Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol 2014;132:1453-60.
5. American College of Rheumatology. Screening for hydroxychloroquine retinopathy. Committee on Rheumatologic Care, 2016.



