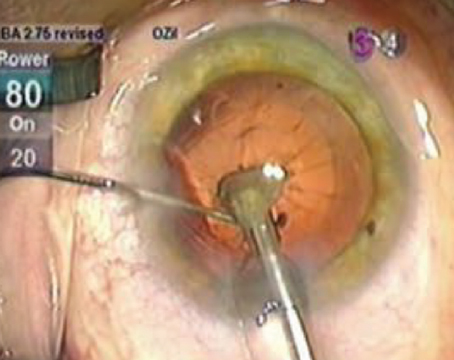
Endoscopic cyclophotocoagulation has been available to ophthalmologists for a number of years. In cases of refractory glaucoma, ECP represents an advance over older transscleral cyclophotocoagulation for cyclodestruction of the ciliary processes. By providing direct visualization not available in TCP, ECP enables treatment with less damage to adjacent tissue.
 |
 |
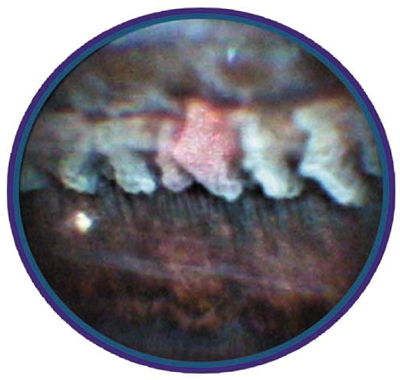
| The desired tissue effect in endoscopic cyclophotocoagulation is to whiten and shrink the ciliary processes. |
Another use of ECP that's been an option for some time is phacoemulsification combined with ECP for cataract patients with medically controlled glaucoma. The benefit of this combined procedure is both lowering of intraocular pressure and decreasing the number of medications needed to maintain adequate IOP control. While many cataract surgeons have successfully used phaco/ECP in this setting, many more have been awaiting longer-term studies and data to support its clinical efficacy, safety and benefit.
In this article, I will share data from studies in which I and some colleagues have participated that I believe may lead to a significant shift in the way we think about cataract surgery in patients with medically controlled glaucoma.
ECP/Phaco Study
My colleagues and I conducted a study to delineate the natural history of phaco in the setting of medically controlled glaucoma, and to determine if adding ECP to phaco results in significantly lower long-term IOP, fewer glaucoma meds, or both when compared to phaco alone. In addition, the study sought to determine if adding ECP to phaco increases risk compared to phaco alone, and to calculate the cost benefits, if any, resulting from long-term decrease in glaucoma medications.
We recently completed data analysis on the prospective, randomized four-year study of 707 total eyes with medically controlled glaucoma that were undergoing cataract surgery; 626 were randomized to ECP/phaco and 81 were randomized to phaco alone. The surgery was performed by five surgeons within the same group. Data on visual acuity, intraocular pressure, medications and complications were collected. The mean follow up was 3.2 years, (range from 0.5 to 5.8 years), extending from January 2000 to December 2004. All patients received a foldable, three-piece silicone IOL.
The parameters of the typical cataract surgery are unchanged in combined phaco/ECP. The anesthesia used can be topical, intracameral or peribulbar block. The incision used can be scleral tunnel or clear corneal. The eye is inflated with viscoelastic and the endolaser probe is inserted through the phaco wound; the ciliary process is visualized and treated directly. The laser endoscope is a 20-ga. probe, and the action is at the tip. It incorporates a very bright, variable xenon light source, a diode laser and a fiber-optic video image. This is connected to a control box.
The ECP treatment zone can extend from 180 degrees to 360 degrees. In our study all of the patients were treated from 200 degrees to 270 degrees. The desired tissue effect is to whiten and shrink the ciliary processes.
Postop medications are also straightforward: In our study, the postop regimens were the same for the phaco/ ECP group as for the phaco alone. All patients received Zymar, Acular and Pred Forte q.i.d.
All patients were initially maintained on their preop glaucoma medications, and if IOP reduction was believed to be adequate based on the patient's optic nerve and visual field, one glaucoma medication at a time was eliminated.
The Results
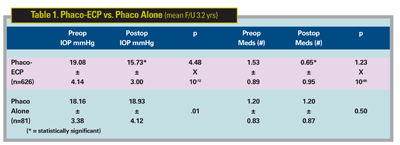
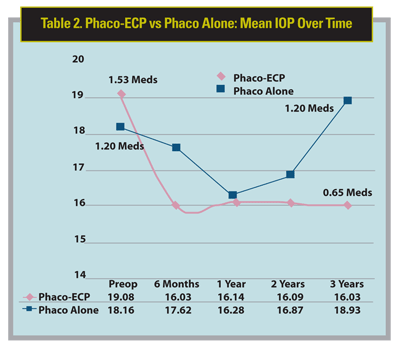
The outcomes we looked at were change in IOP and stability over the follow-up period, and change in number of glaucoma medications compared to preop. (See Tables 1 and 2 for the results).
In the 626 combined phaco/ECP eyes, the preop IOP was 19.08 mmHg and at the end of the study period of 3.2 years on average, the postop IOP was 15.73; this was highly statistically significant with a p value of 10-72.
The number of preop medications in the phaco/ECP group was 1.53; this was dramatically reduced at the end of the study period to 0.65, and again this was highly statistically significant with a p value of 10-85.
The phaco alone group included 81 eyes. The preop IOP was 18.16 mmHg; and at the end of the study period, it was 18.93. The number of preop medications in the phaco alone group was 1.2 and it remained 1.2 at the end of the study.
In the phaco/ECP group, in 68 percent there was a decrease in medications, in 27 percent there was no change in medications, and in 5 percent there was an increase in medications. This compares to the phaco alone group, wherein only 11 percent of eyes experienced a decrease in medications. In 77 percent there was no change in medications, and in 12 percent there was an increase in medications.
Another important result is the percentage of patients who were on two or more glaucoma medications in the two groups. In the phaco/ECP group, preoperatively there were 44 percent; postoperatively, this fell to 18 percent and was highly statistically significant. In the phaco alone group, 32 percent of eyes were on two or more medications preop, and at the end of the postop period, 38 percent were on two or more medications.
A vital consideration is the percentage of subjects with a reduction in IOP at the end of follow up. We know that cataract surgery alone may reduce IOP in some glaucoma patients. One of the key parameters, therefore, in considering a combined procedure is how it compares over time with cataract surgery alone.
—S. Berke, MD |
In the phaco/ECP group there was a decrease in IOP in 79 percent of eyes, no change in 9 percent and an increase in 12 percent at the end of follow-up. In the phaco alone group, however, there was a decrease in IOP in 38 percent, no change in 2 percent and an increase in 60 percent.
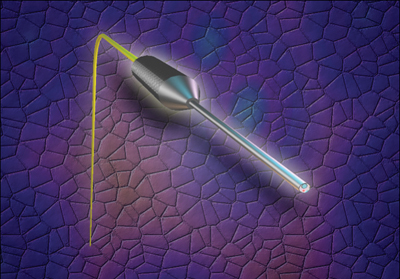  |
| The 20-ga. laser endoscope incorporates a very bright, variable xenon light source, a diode laser and a fiber-optic video image. |
Complications, Cost Issues
No serious complications were reported in either group. The incidence of CME was equal in both groups at approximately 1 percent. (For additional information on a larger study of ECP complications, see sidebar, p. 68.)
The average annual cost of glaucoma medications for patients in the phaco/ECP group was $2,641 preop and $1,137 postop. This resulted in an average annual savings of $1,504 for each patient in the phaco/ECP group. The phaco alone group did not experience any financial savings for glaucoma medications.
In summary, this long-term analysis of medically controlled glaucoma patients undergoing cataract surgery demonstrates that phaco/ECP results in significantly lower IOP and significantly fewer medications compared to phaco alone, without any increase in early or late complications.
The statistical results of this study are sufficiently powerful to suggest a paradigm shift in our thinking about treatment of cataract patients with concurrent glaucoma. It is evident from this study that phaco alone does not contribute to long-term IOP control, while adding ECP to phaco does. The high safety margin of ECP in this large cohort with extended follow up clearly indicates that this method of rendering decreased outflow is benign. Combining ECP with phaco should be considered in patients with cataracts and medically controlled glaucoma.
Dr. Berke is an associate clinical professor of ophthalmology and visual sciences at Albert Einstein College of Medicine, chief of the Glaucoma Service at Nassau University Medical Center, and in private practice at Ophthalmic Consultants of Long Island. Contact him at sberke@ocli.net.